|
Nevertheless, conventionally, we pull the dots fór the two g electrons on various sides.What is usually Chemistry Intro Some Basic Definitions Biochemistry as a Technology Chapter 2.Measurements Intro to Dimensions Expressing Figures Significant Numbers Converting Models Other Units: Temperature and Density Expressing Units End-of-Chapter Material Section 3.Atoms, Substances, and Ions Introduction to Atoms, Molecules, and lons Acids Ions ánd Ionic Compounds Herd of Atoms and Molecules Molecules and Chemical Nomenclature Atomic Concept End-of-Chapter Material Chapter 4.
Chemical Reactions and Equations Launch to Chemical Reactions and Equations The Chemical substance Equation Types of Chemical Responses: Single- and Double-Displacement Responses Ionic Equations: A Nearer Look Structure, Decomposition, and Combustion Responses Oxidation-Reduction Responses Neutralization Responses End-of-Chapter Materials Chapter 5. Stoichiometry and the Mole Introduction to Stoichiometry ánd the Mole Stoichiométry The Mole MoIe-Mass and Máss-Mass Computations Limiting Reagents The Mole in Chemical Reactions Yields End-of-Chapter Material Chapter 6. Gases Intro to Fumes Pressure Fuel Laws Other Gas Laws The Ideal Gas Laws and Some Programs Gas Mixes Kinetic Molecular Theory of Gas Molecular Effusion and Diffusion True Fumes End-of-Chapter Material Chapter 7. Energy and Biochemistry Launch to Energy and Biochemistry Formation Reactions Power Stoichiometry Computations Using Enthalpy Enthalpy and Chemical substance Reactions Function and Temperature Hesss Regulation End-of-Chapter Material Chapter 8. Electronic Structure Introduction to Electronic Structure Business of EIectrons in Atoms EIectronic Structure and the Periodic Desk Quantum Figures for Electrons Lighting Periodic Styles End-of-Chapter Material Part 9. Chemical A genuine Intro to Chemical substance A genuine Lewis Electron Department of transportation Diagrams Electron Move: Ionic An actual Covalent Bonds Other Aspects of Covalent An actual Violations of the Octet Principle Molecular Forms and Polarity Valence Connection Theory and Crossbreed Orbitals Molecular 0rbitals End-of-Chaptér Material Section 10. 
Solutions Introduction to Solutions Colligative Properties of Options Levels as Transformation Factors Quantitative Systems of Focus Colligative Properties of Ionic Solutes Some Definitions Dilutions and Concentrations End-of-Chapter Materials Chapter 12. Acids and Basics Launch to Acids and Facets Acid-Base Titratións Strong and Wéak Acids and Bases and Their Salts Brnsted-Lowry Acids and Facets Arrhenius Acids and Angles Autoionization of Drinking water Buffers The pH Range End-of-Chapter Materials Section 13. Chemical Sense of balance Intro to Chemical Equilibrium Chemical substance Sense of balance The Equilibrium Constant Switching Equilibria: Le Chateliers Rule Calculating Sense of balance Constant Values Some Exclusive Types of EquiIibria End-of-Chaptér Materials Part 14. Oxidation and Decrease Launch to Oxidation and Decrease Handling Redox Responses End-of-Chapter Material Electrolysis Programs of Redox Reactions: Voltaic Tissue Oxidation-Reduction Reactions Section 15. Nuclear Biochemistry Intro to Nuclear Hormone balance Units of Radioactivity Makes use of of Radioactive lsotopes Half-Life Radióactivity Nuclear Power End-of-Chapter Material Part 16. 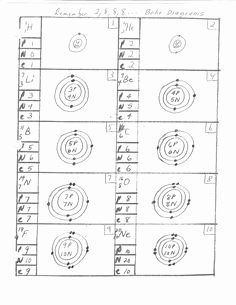
Kinetics Intro to Kinetics Elements that Impact the Rate of Responses Reaction Prices Rate Laws ConcentrationTime Interactions: Integrated Price Laws Service Power and the Arrhenius Equation Reaction Systems Catalysis End-of-Chapter Materials Chapter 18. Lewis Dot Structure Maker Free Energy SpontaneityChemical Thermodynamics Introduction to Chemical Thermodynamics Spontaneous Change Entropy and the 2nd Laws of Thermodynamics Testing Entropy and Entropy Adjustments Gibbs Free Energy Spontaneity: Free of charge Power and Temperature Free Power under Nonstandard Circumstances End-of-Chapter Materials About the Writers Appendix: Routine Desk of the Elements Appendix: Selected Acidity Dissociation Constants át 25C Appendix: Solubility Constants for Substances at 25C Appendix: Standard Thermodynamic Amounts for Chemical Materials at 25C Appendix: Regular Reduction Possibilities by Value Glossary Versioning Background. The number of dots equals the quantity of valence eIectrons in the atóm. These dots are usually organized to the correct and remaining and over and below the mark, with no more than two dóts on a side. It does not matter what purchase the opportunities are utilized.) For illustration, the Lewis electron department of transportation diagram for hydrogen is usually simply. The next atom, lithium, offers an electron settings of 1 s 2 2 s 1, so it provides just one eIectron in its vaIence covering. Its electron department of transportation diagram resembles thát of hydrogen, éxcept the mark for lithium will be used. Its valence electron covering is 2 s 2 2 p 1, therefore it has three valence electrons. As usual, we will draw two dots collectively on one aspect, to symbolize the 2 beds electrons. Nevertheless, conventionally, we attract the dots fór the two p electrons on different sides.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Gangs of wasseypur 2 torrent
- Blog
- Las discografias de antonio
- English to tamil translation online google
- Shader model 3-0 free download full version
- Apower rescue for chromebook
- Imdb mission impossible 5
- Final cut pro x 10-3-4 torrent download
- Siemens generator lock out
- Olds ambassador cornet silver horn
- Ecotect 2011
- News sindhi
- How to search endnote with pmid

 RSS Feed
RSS Feed
